The Science of Where Drugs Go
Similar to the fields of Pharmacology and Toxicology, ADME/Drug Metabolism is a well-established field dating back to the nineteenth century, and it is very complex with respect to the wealth and depth of information available. It is still evolving today. The goal of this post is not to address every detail of the field, but instead to give readers a basic introductory understanding of the discipline. Further details about the many aspects of ADME/Drug Metabolism can be accessed online, or in scientific journals.
In my Pharmacology and Toxicology posts, I briefly discussed Pharmacokinetics, Toxicokinetics, and the Absorption, Distribution, Metabolism and Excretion (ADME) of drugs and other xenobiotics. These areas collectively comprise the exciting field of “Drug Metabolism”. Whenever a new drug or industrial chemical is produced, several key aspects of the chemical must be characterized; how much of it gets absorbed into the body, where it goes in the body, and how long it stays there. The answers to these questions are collectively known as the molecule’s “ADME” profile, and a tremendous amount of work goes into this type of research. It’s very critical information as it helps characterize the chemical’s subsequent pharmacological or toxicological effectiveness and properties. As you read through this post keep a couple of key questions in mind. How much of the molecule gets absorbed into the body? Where does it go once absorbed? How long does it stay in the body? Is it transformed into something new? How does it leave the body?
The Liver, Kidney and Small Intestine
Before walking through the ADME acronym in greater detail lets first talk about the three organs that impact a chemical’s ADME profile the most: the Liver, the Kidneys and the Small Intestine. As described later in this post, other organs can impact a chemical’s ADME profile but these are the three major players. I will try to explain these organs in the context of this post in the simplest way possible.
“If you take a pill and it simply sits in your stomach for a brief period of time, and then passes out through your feces, then it technically wasn’t absorbed into your body,” said the same professor from my Pharmacology post who distinguished the discipline of Pharmacology from Pharmacy. This anecdote described how drugs and man-made industrial chemicals in the classic sense must be absorbed into the blood stream to actually have been absorbed into your body. There are other ways a chemical can get into the body (inhalation and dermal exposures), but for this post I’m focusing only on oral absorption.
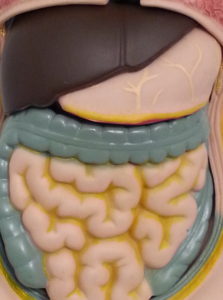
If absorbed in the small intestine, the molecule of interest then passes through the “Hepatic Portal Vein” into the liver where any number of things can happen to it (described in greater detail below). After leaving the liver and entering the general circulation, the molecule is for the most part cleared through the kidneys via the urine, but in some cases it can be deposited back into the GI-Tract and leave the body through the feces. Molecules can also be exhaled depending upon what they’re transformed into once absorbed. Any molecule not absorbed by the small intestine leaves the body through the feces.
I can’t emphasize enough the importance of the liver and kidneys which both perform numerous critical functions in the body. For this particular context, the normal function of both, are critical to the body’s handling of both endogenous and exogenous chemicals which is why physicians, nurses and pharmacists inquire about their function early on when patients are admitted for care in clinical settings.
Breaking Down the Acronym
With that in mind, I’ll now break down the ADME acronym and why these skill sets are so valuable for scientists who gain an expertise in them:
- Absorption: As described earlier, the “Absorption” aspect deals with how much of the chemical is absorbed into the body following oral ingestion, passage into the small intestine, the liver, and finally into the blood stream. The properties of the chemical itself can dictate how much of it is absorbed – particularly its size and for simplicity whether it’s charged (+ or -) or neutral. As described in my Pharmacology post, pharmaceutical companies designate molecules as either “small” or “large”, and many large molecules can only be administered by injection into the bloodstream. While there are several experiments that can help characterize a molecule’s oral absorption, “Pharmacokinetic” and “Biliary Excretion” studies (discussed below) are the most specific. Just briefly, by treating animals (usually rodents) with radio-labelled compound, the amount of compound absorbed can be determined by quantifying the amount of radioactivity in the blood, urine and feces over time telling scientists how quickly the molecule was absorbed in addition to the amount absorbed and if these two metrics change with increasing dose.
 Metabolism: Once in the body, molecules can undergo “Biotransformation” – that is, classes of proteins called “Enzymes” can transform the structure of a given molecule by breaking it down into multiple pieces or adding on new “Functional Groups”, altering its properties so that it’s more readily eliminated from the body (discussed below). In some instances, this biotransformation turns the molecule into something toxic which can cause injury to the liver or other tissues in the body. The Metabolism aspect of ADME, involves a separate discipline called “Enzymology” which focuses just on the enzymes themselves; their levels in cells (Protein Expression), the rates of their reactions (Kinetics), their structures, etc. There are actually multiple classes of drug metabolizing enzymes but the most prevalent class at least as it relates to the liver, is the “Cytochrome-P450s”. Pharmaceutical companies pay particular attention to this class of enzyme (and a host of others) as they greatly impact the “Bioavailability” of the drug. The “First Pass Effect” or “First Pass Metabolism” occurs when a drug is significantly metabolized before it gets into the general circulation due to metabolism by liver enzymes. Some of the clinical aspects of metabolism will be further discussed later in this post. By the way, while Cytochrome P450s were classically associated with the liver, we now know that they are expressed throughout the human body as well as all plants and animals.
Metabolism: Once in the body, molecules can undergo “Biotransformation” – that is, classes of proteins called “Enzymes” can transform the structure of a given molecule by breaking it down into multiple pieces or adding on new “Functional Groups”, altering its properties so that it’s more readily eliminated from the body (discussed below). In some instances, this biotransformation turns the molecule into something toxic which can cause injury to the liver or other tissues in the body. The Metabolism aspect of ADME, involves a separate discipline called “Enzymology” which focuses just on the enzymes themselves; their levels in cells (Protein Expression), the rates of their reactions (Kinetics), their structures, etc. There are actually multiple classes of drug metabolizing enzymes but the most prevalent class at least as it relates to the liver, is the “Cytochrome-P450s”. Pharmaceutical companies pay particular attention to this class of enzyme (and a host of others) as they greatly impact the “Bioavailability” of the drug. The “First Pass Effect” or “First Pass Metabolism” occurs when a drug is significantly metabolized before it gets into the general circulation due to metabolism by liver enzymes. Some of the clinical aspects of metabolism will be further discussed later in this post. By the way, while Cytochrome P450s were classically associated with the liver, we now know that they are expressed throughout the human body as well as all plants and animals.- Distribution: Once in the body’s general circulation, the molecule can travel to many of the tissues of the body and can accumulate there for short- or long-periods of time depending upon the tissue and the properties of the molecule itself. If the molecule is particularly non-polar (neutral), it can accumulate in fatty tissues or for pregnant females, it can partition into breast milk and be transfered to nursing offspring. Molecules can also bind reversibly to blood plasma allowing for an increased internal dose.
 Excretion: Excretion refers to how the molecule is eliminated from the body. Typically the urine and the feces are measured to determine how the molecule is eliminated. Detection in the urine indicates that the molecule was absorbed to some degree into the bloodstream as the kidneys filter out aqueous waste from the blood. Poor kidney function can actually lead to a prolonged bioavailability and subsequent toxicity which is why clinicians always inquire about it as described earlier. Not all of the absorbed chemical exits the body through the urine though. It turns out that absorbed chemicals can empty out back into the GI-Tract from the liver via the bile and then be eliminated through the feces.
Excretion: Excretion refers to how the molecule is eliminated from the body. Typically the urine and the feces are measured to determine how the molecule is eliminated. Detection in the urine indicates that the molecule was absorbed to some degree into the bloodstream as the kidneys filter out aqueous waste from the blood. Poor kidney function can actually lead to a prolonged bioavailability and subsequent toxicity which is why clinicians always inquire about it as described earlier. Not all of the absorbed chemical exits the body through the urine though. It turns out that absorbed chemicals can empty out back into the GI-Tract from the liver via the bile and then be eliminated through the feces.- Drug Transport: This aspect doesn’t traditionally fall under the ADME acronym, but it’s an important field that is now being actively researched in academia and industry. It deals with how cells may concentrate the chemical in tissues or remove the molecule from the target tissue before it can exert its function.
- Pharmacogenomics/Toxicogenomics: These new and exciting fields look at the genetics unique to individuals to determine the best treatments and dosages for that individual. Genetic differences in levels in the drug metabolizing enzymes mentioned above can result in drastically different effects of treatment with a given dose of a drug depending on the individual. The same is true for an individual’s reaction to a toxicant.
The Applications for ADME

So why is all of this important? Whether in a hospital setting, a pharmacy, or in the chemical industry creating a new food additive, pesticide, or cosmetic, it’s important to have as clear an understanding as possible of where the molecule goes in the body and what its fate is following ingestion. As described above, physicians, whether in general practice or in the emergency room, have to gauge a patient’s liver and kidney function as those two organs will dictate how long the pharmaceutical stays in circulation – again, its “Bioavailability”.
Even a pharmaceutical designed to be therapeutically beneficial can be toxic if it remains in the body too long, if its levels exceed a certain dose level, if it’s transformed into something toxic, or if there is a drug-drug interaction. “Drug-Drug Interactions” are typically the result of one drug causing an increased internal dose of another drug due to inhibiting or preventing metabolism by the enzymes described in the metabolism bullet above. In graduate school we learned about the classic case of Terfenadine causing abnormal heart rhythms that could lead to death by increasing the amounts of circulating Erythromycin – both of these drug molecules normally work by with non-cardiac mechanisms. Terfenadine was removed from the market once it’s ability to cause this deadly drug-drug interaction was recognized.
Where ADME is Considered
Chemical, food and beverage, and pharmaceutical companies all have to know what happens to their molecules in the body for several reasons. A drug can be highly effective at preventing cancer cells from multiplying in a laboratory setting in dishes and flasks, but unless it is readily absorbed in the intestines and can actually get to its target tissue in the body as its untransformed structure, it’s useless. In some instances, a drug can get to its site of action, but the cells of that tissue can adapt and effectively expel the molecule before it gets a chance to exert its function as described earlier. Pesticides which are sprayed on agricultural commodities often make it to our dinner tables in low levels where we do ingest them to some degree.

Another very important context for ADME/Drug Metabolism is actually “Food Safety” which is a key consideration for food and beverage companies like Pepsico and Quaker Oats. As a matter of fact, at a family dinner earlier this year, a discussion of about Trisodium Phosphate (TSP), a preservative used in “Cap’n Crunch” cereal which had other industrial uses, caused a stir amongst my relatives. I had to remind them that both the ADME and toxicity profiles of this preservative had already likely been characterized and cleared through extensive studies by the company and the Food and Drug Administration (FDA).
ADME Careers and Training
We’ve discussed what ADME/Drug Metabolism is, but where do these scientists work and where do they receive their training? ADME/Drug Metabolism scientists work in Pharmaceutical and Chemical companies, and in government performing “Regulatory” functions (visited in an upcoming post). They receive their training for the most part in academic settings in labs specializing in Pharmacology and Toxicology, both of which have ADME/Drug Metabolism as a major component. There are some labs that strictly study one of the many aspects of ADME/Drug Metabolism but they are in the minority of the research groups in the biomedical sciences.
“Scientists with training in Drug Metabolism will almost never be with jobs,” said one of the professors in my graduate department at the University of Michigan. While we know that there is no job that is 100% secure, this particular professor was stressing that ADME/Drug Metabolism scientists are critical parts of most companies. The divisions of those companies responsible for these types of studies are typically titled Drug Metabolism and Pharmacokinetics (DMPK). No matter what disease the company is interested in (Diabetes, Cancer, HIV, etc.), it is essential that they understand the chemical’s ADME/Drug Metabolism profile for their own purposes and when submitting packages for approval by the Food and Drug Administration and other regulatory agencies. The same is true for chemical companies. Depending on the degree level earned and where the scientist is employed, ADME/Drug Metabolism scientists can earn starting salaries of $60,000-$70,000.
Technologies Used in ADME Experiments
There are numerous scientific tools and technologies that ADME/Drug Metabolism scientists use, but I’ll mention two of them briefly. The first is the Mass Spectrometer also known as the “Mass-Spec”. The accompanying picture shows a Mass-Spec. Click on the image to enlarge it. Mass-Specs are not used solely in ADME/Drug Metabolism studies, but they’re very important to the field because they can detect and identify molecules in whole blood, blood plasma, tissue samples, urine, and fecal samples at very, very low levels. More importantly they can detect changes in the structure and identity of molecules once they have gone through the body and can help to predict a drug/industrial chemical’s efficacy or toxicity.
Technologies and methods are always changing and evolving but the Mass-Spec is currently a very important tool for ADME/Drug Metabolism. Currently, Pharmacology and Toxicology scientists in industry are moving towards decreasing animal usage and towards more in vitro and in silico methods which are giving rise to the use of Physiologically Based Pharmacokinetic Models (PBPK) where the fate of molecules can be predicted using various constants and inputs into computational models. We’re currently in the early era of these methods.
Professional Societies for ADME
If you are interested in learning more about the exciting field ADME/Drug Metabolism, I suggest that you visit the website of the American Society for Pharmacology and Experimental Therapeutics (ASPET). You can then click on the link titled Education & Careers. In the right hand column, there is a link titled About Pharmacology, that provides a great deal of interesting information. Speaking of ASPET, all scientific disciplines have their own professional societies with annual meetings that are held in various cities (eg. Boston, San Francisco, Chicago, San Diego, Washington,etc.) every year, and where scientists gather to show their results and network. The two major professional societies for ADME/Drug Metabolism scientists are ASPET, and the International Society for the Study of Xenobiotics (ISSX).
Thank you for taking the time to read this post, and I hope I was able to shed some light onto what ADME/Drug Metabolism is as a field. The next post in this series will discuss the field of Inhalation Toxicology. If you enjoyed this post, you may also enjoy:
• A look at STEM: What is Pharmacology?
• A look at STEM: What is Toxicology?
• A look at STEM: What is Inhalation Toxicology?
• A look at STEM: Blockchain technology, a new a conducting business and record keeping
A special thank you is extended to Dr. Paul Hollenberg and Dr. Chester Rodriguez for their contributions to this post. I also want to acknowledge Dr. Yoichi Osawa of the University of Michigan’s Department of Pharmacology for the picture of the Mass Spectrometer used in this post.
The Big Words LLC Newsletter
For the next phase of my writing journey, I’m starting a monthly newsletter for my writing and video content creation company, the Big Words LLC. In it, I plan to share inspirational words, pieces from this blog and my writers blog, and select videos from my four YouTube channels. Finally, I will share updates for my book project The Engineers: A Western New York Basketball Story. Your personal information and privacy will be protected. Click this link and register using the sign-up button at the bottom of the announcement. If there is some issue signing up using the link provided, you can also email me at bwllcnl@gmail.com . Best Regards.


Journal of Pharmacokinetics & Experimental Therapeutics is an open access journal that focuses on pharmacokinetic properties of drugs, including Absorption, Distribution, Metabolism and Excretion (ADME) while underlining the importance of experimental therapeutics in pharmacokinetic research. Journal of Pharmacokinetics & Experimental Therapeutics accepts manuscripts in clinical pharmacokinetics, population pharmacokinetics, antibody pharmacokinetics, pharmacokinetics review, vancomycin pharmacokinetics, tacrolimus pharmacokinetics, pharmacokinetic drugs, pharmacokinetics intravenous, pharmacokinetics oral, pharmacokinetics renal, pharmacokinetics human, pharmacokinetics rat, pharmacokinetics mice, pharmacokinetics for children, pharmacokinetics research to promote healthy life are welcome.
If your interested can submit your manuscript.
That manuscript should not published in any other journal pages.
You can submit your manuscript, to the below email id.
Email id: pharmacokinetics23@gmail.com
Journal url:
https://www.omicsonline.org/pharmacokinetics-and-experimental-therapeutics.php